Research Fields > Skin (Dermatology)
Unraveling the Mystery of the Infant Skin Barrier From the Principles of Infant Skin Development to the Biomimetic Technology Revolution - Developing Artificial Infant Skin Using iPS Cell Technology
Pigeon endeavors to enhance the health of infant skin, which has a vulnerable barrier function to prevent allergic march. Introducing Pigeon’s development of artificial infant skin using iPS cell technology.
Protecting the Most Delicate Protective Barrier: A Scientific Breakthrough in Baby Skincare
Infant skin is only one-third as thick as adult skin, and the underdeveloped barrier function of infant skin puts infants at high risk for conditions such as eczema. However, conventional skincare product development has relied on adult data and animal testing, which overlooks the differences in the essential characteristics of infant skin. Seeking to develop specialist products that supplement the barrier function of immature infant skin, Pigeon conducted three studies and used the results to build a comprehensive solution covering everything from basic mechanisms to applied technology.
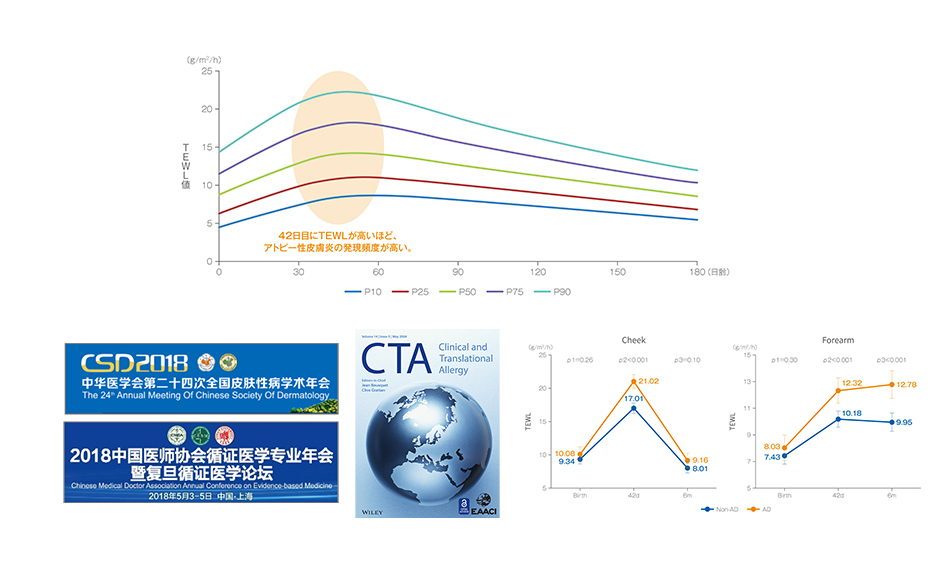
Study 1: Deciphering the infant skin development map from China – Research on the skin barrier function in infants
Study 2: The natural repair code hidden in the vernix caseosa – A new mechanism by which vernix caseosa lipids regulate the skin barrier function
Study 3: Technological revolution in baby skincare product evaluation – Developing artificial infant skin using iPS cell technology
Study 3: Key Points
This page explains Study 3 from the list above.
- Pigeon focused on iPS cells and succeeded in developing artificial infant skin with a stratum corneum similar to that of living skin.
- The artificial infant skin developed by Pigeon is expected to solve cell shortages, improve experimental accuracy, replace animal testing, and be applied to personalized medicine.
What Pigeon Aimed to Achieve through Artificial Infant Skin Research
Unique structure of infant skin: The structure of infant skin differs from that of adults. This means that a completely distinct approach is required to develop baby products.
Problems with existing experimental methods: When developing cosmetics and medical products, testing for skin irritation is an essential process. However, existing approaches like animal testing and infant skin raised issues around ethics and physiological applicability. The same is true for developing baby skincare products.
Solutions offered by artificial infant skin: Pigeon’s objective was to develop artificial infant skin that would meet requirements around safety, effectiveness, and ethicality in the baby skincare development process, to overcome these issues. This would allow safe and effective product testing and evaluation in an environment like that of actual infant skin.
This research and development of artificial infant skin is an important step for developing skincare products that can be used on babies effectively and with peace of mind. Pigeon has already introduced tests using artificial infant skin into some areas of product development, and plans to expand this coverage to other items in its product lineup in the future.
Key Points of the Artificial Infant Skin Developed by Pigeon
Key points in the development process were as follows.
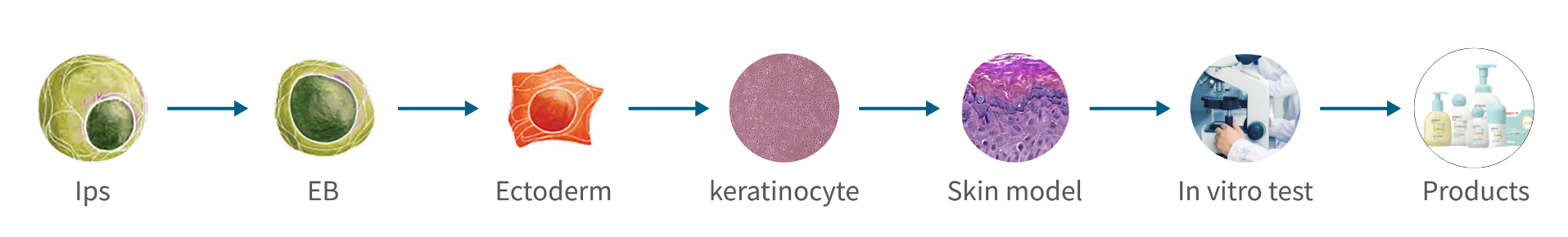
1.Use of iPS cells
iPS cells can transform into any cell in the body and are also known as “age-reversed cells.” Pigeon sought to use these cells to artificially create a skin model closely resembling actual infant skin.
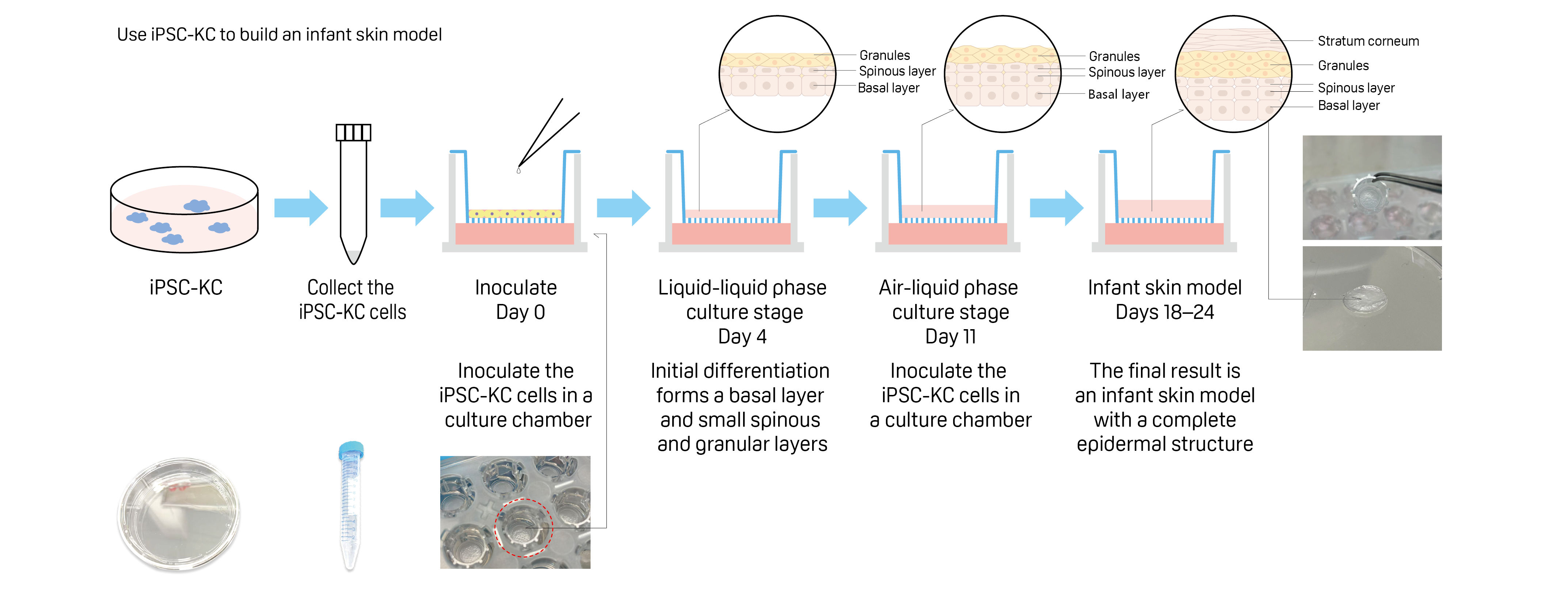
2.Optimized cultivation process
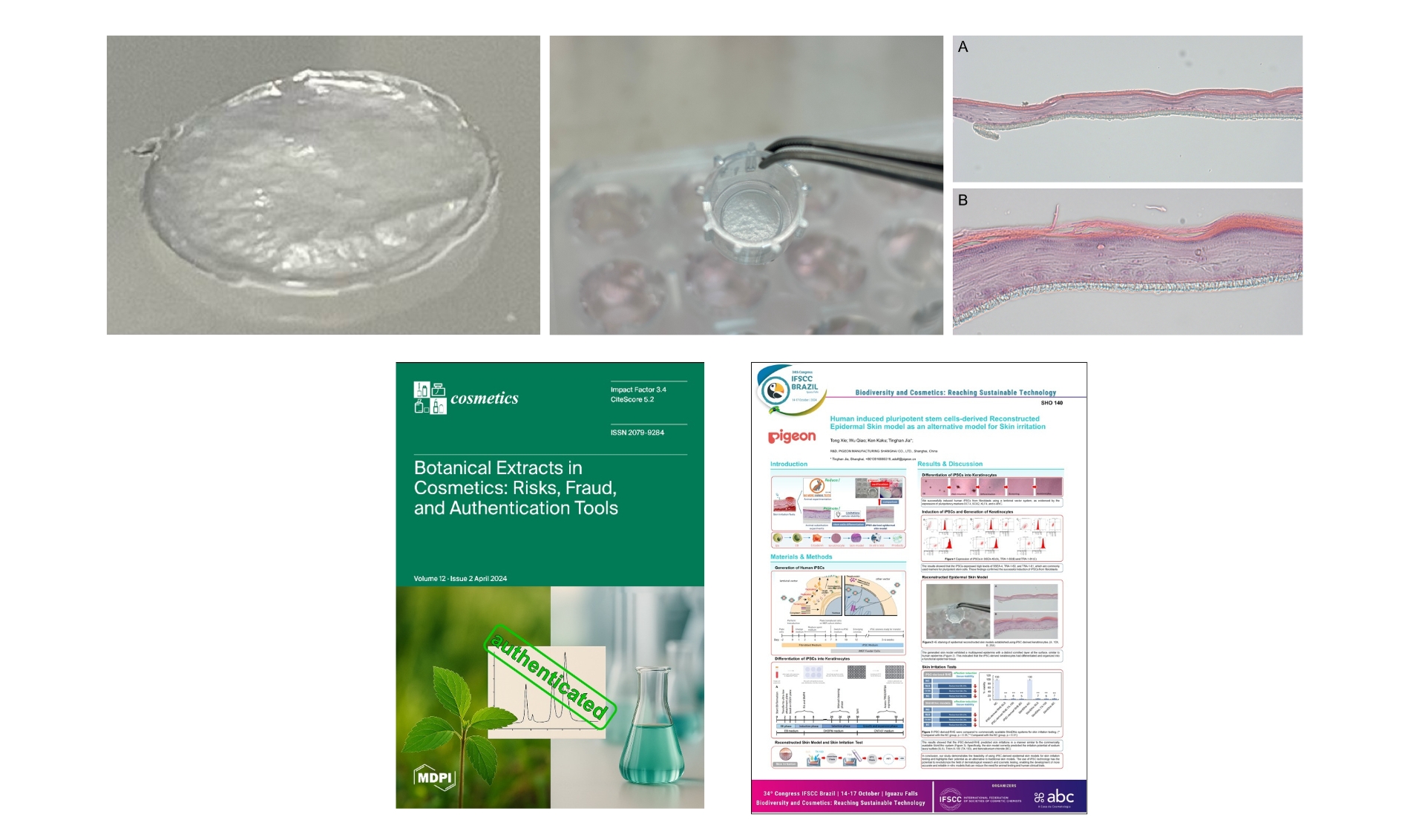
By improving the culturing method, Pigeon successfully created an artificial skin model that included the stratum corneum, which has the skin barrier function, within 24 days, which is faster than usual. (Fig. 1A)
3.Functional improvements over existing models
The new artificial infant skin model demonstrated the following superior characteristics compared to traditional skin models:
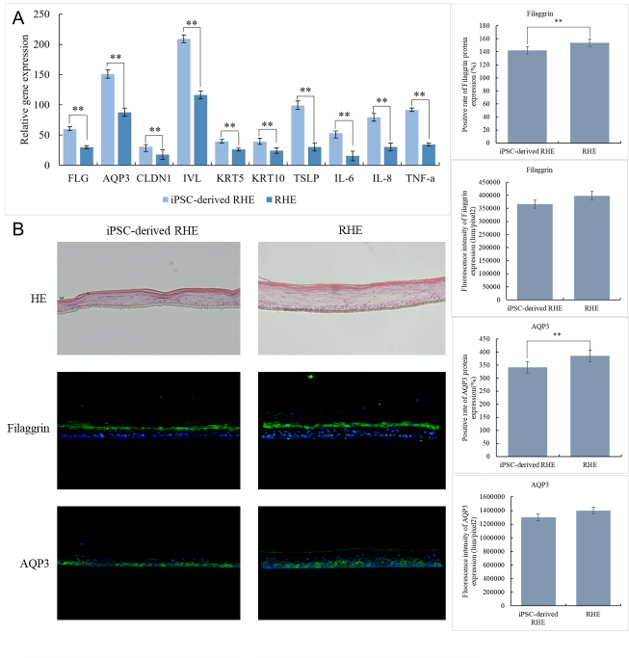
・High barrier function and metabolic activity:
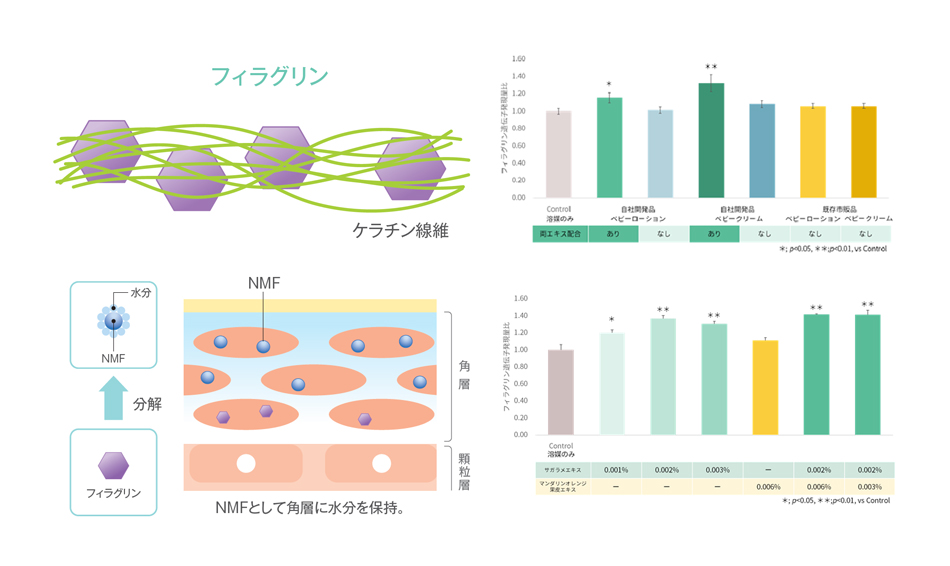
The expression level of important genes involved in skin function, including aquaporin 3 (AQP3), the barrier protein (CLDN1), and keratin (KRT10), was found to be meaningfully higher than in the traditional model using normal human epidermal keratinocyte cells (NHK-derived RHE). This indicates that iPS cell (IPSC)–derived RHE has extremely high vitality and regenerative capacity. (Fig. 1B)
・Realistic stress response:
Functionality equivalent to that of the standard international model (SkinEthicTM) was shown, and it was possible to accurately judge the level of irritation (stress experienced by actual skin) caused by the chemical substances tested: sodium laurel sulfate (SLS), Triton X-100 (TX-100), benzalkonium chloride (BC), etc.
Construction and evaluation of infant skin model using iPSC-derived keratinocytes (iPSC-KCs)
Fig. 1. (A) Gene expression in iPSC-derived RHE and RHE, (B) HE chromosome results and Filaggrin/AQP3, (C) Immunofluorescence staining results
Impact of the Artificial Infant Skin Developed by Pigeon on the Development Environment
The artificial infant skin developed by Pigeon is a significant advance in safety evaluation during the research and development of cosmetics and pharmaceuticals and is expected to have a major impact on the development environment by resolving issues such as cell shortages.
・Resolution of cell shortages
Because iPS cells can proliferate indefinitely, therefore, they can help solve challenges around cell donor shortages and stability of supply.
・Improved experimental accuracy
Because skin models with identical genetic backgrounds can be mass-produced, variability in experimental results caused by individual differences can be eliminated, enabling the acquisition of more accurate data.
・Replacement for animal testing
This model offers a highly accurate replacement for animal testing in cosmetic safety evaluations. This accelerates the development of animal-free products and represents significant progress for animal welfare.
・Application to personalized medicine
This technology enables the creation of artificial skin from the iPS cells of people with specific skin diseases, such as atopic dermatitis. Its expected applications include elucidating disease mechanisms and personalized medicine/skincare—the development of treatments and drugs tailored to the individual.
Comment from Pigeon Researcher
This is the first research applying iPS artificial pluripotent stem cell technology to the structure of artificial infant skin. Infants have a thinner stratum corneum than adults, and the skin barrier function is immature. Their skin has a high moisture content, but it dries easily, and their cells regenerate rapidly. The artificial infant skin model that we constructed (iPSC-derived RHE) can simulate these characteristics of infant skin extremely well. I hope that the results of this research will be used in baby skincare development and contribute to further research on infant skin.
Bibliographical Information
Paper Title: Human Induced Pluripotent Stem Cells-Derived Reconstructed Epidermal Skin Model as an Alternative Model for Skin Irritation
Authors: Tong Xie, Wu Qiao, Tinghan Jia and Ken Kaku
URL: https://doi.org/10.3390/cosmetics12020075
Journal: Cosmetics 2025, 12(2), 75
General Bibliography
1)https://www.163.com/dy/article/FIIO4I330514D84S.html
2)https://www.episkin.com/skin-irritation
3)Xie, T.; Qiao, W.; Jia, T.; Kaku, K. Human Induced Pluripotent Stem Cells-Derived Reconstructed Epidermal Skin Model as an Alternative Model for Skin Irritation. Cosmetics 2025, 12, 75. https://doi.org/10.3390/cosmetics12020075
Papers and Presentations
関連記事
- Home
- Research and Development
- Research Fields
- Skin (Dermatology)
- Unraveling the Mystery of the Infant Skin Barrier From the Principles of Infant Skin Development to the Biomimetic Technology Revolution - Developing Artificial Infant Skin Using iPS Cell Technology